|
||||||||||
|
||||||||||
| Favorite Tweets by @thaibirding | ||||||||||
|
||||||||||
 Buy Birds of Thailand on Amazon.co.uk |
||||||||||
| Site Map ; Contributors |
| An
analysis of records of three passage migrants in Thailand: Tiger Shrike
Lanius tigrinus, Yellow-rumped Flycatcher Ficedula zanthopygia
and Mugimaki Flycatcher F. mugimaki By Philip D. Round |
||||||||||||||||||||||||||||||||||||||
Tweet
 |
||||||||||||||||||||||||||||||||||||||
| Records of three passage migrants in Thailand are analysed to show seasonal and geographical differences in their distribution. While two of these, Tiger Shrike Lanius tigrinus and Yellow-rumped Flycatcher Ficedula zanthopygia, are early autumn migrants, the third species, Mugimaki Flycatcher F. mugimaki, is a much later autumn migrant and, in contrast to the other two, comprises birds that winter in Thailand as well as passage migrant individuals that winter beyond the country’s southern border. Disproportionately more Mugimaki Flycatchers are recorded on spring migration than the other two species. Definitive-plumaged males of both flycatcher species were recorded earlier on spring and autumn migration than others (grouped female or immature birds). Although in recent decades the numbers of migrants reported have increased due to increased ornithological recording, the numbers of Tiger Shrikes have not increased in proportion to those of Yellow-rumped Flycatchers, possibly indicating a decline in numbers. | ||||||||||||||||||||||||||||||||||||||
| INTRODUCTION In the late 1960s and early 1970s, the journal British Birds presented a series of papers analysing the patterns of occurrence of scarce migrant birds in the British Isles. These were eventually compiled in book form (Sharrock 1974). The clear and straightforward graphical analyses therein were invaluable in giving a much wider and easily comprehensible picture of seasonal occurrence patterns, and differing geographical distributions of the species covered, inspiring ornithologists and providing the stimulus for future fieldwork and data synthesis. Similarly detailed analyses for an Asian site (Hong Kong, probably one of the most heavily watched sites anywhere on the East Asian flyway) were provided by Chalmers (1986) and Carey et al. (2001) who gave seasonal breakdowns of records for most migrant species. Increased field observation and submission of ornithological records for Thailand and other South-East Asian countries are now also beginning to generate datasets large enough to examine better the distribution and seasonality of many migrant birds. Roughly one in three of all bird species in Thailand is at least partly migratory. Precise patterns of occurrence differ widely among species, and in some are undeniably complex. Thailand may be host to three or four discrete populations of (e.g.) Black Bittern Dupetor flavicollis and Chestnut-winged Cuckoo Clamator coromandus: passage migrants, non-breeding winter visitors from countries to the north, summer (wetseason) breeding visitors, and possibly some year-round residents as well (Lekagul & Round 1991). However, the overwhelming majority of migrant species, both landbirds and waterbirds, are non-breeding visitors from countries to the north that spend the Palaearctic winter in tropical South-East Asia. For many of these Thailand marks the southern limit of their South- East Asian winter range. Even here, however, the situation is complicated by the country’s c.14° latitudinal span, and its range of habitats, from seasonally dry monsoon forests to rainforest. Many Palaearctic migrants, although common in ‘continental Thailand’ (north of c.12°N), scarcely enter the Thai-Malay Peninsula (e.g. Siberian Rubythroat Luscinia calliope). Others, e.g. Arctic Warbler Phylloscopus borealis and Eastern Crowned Warbler P. coronatus, appear not to winter much north of c.14° or 15°N, and indeed the peninsular provinces may be their major wintering area in the country. The present paper seeks to examine the patterns of occurrence of three Palaearctic migrant visitors which are unusual in that they occur chiefly as spring and autumn passage migrants: Tiger Shrike Lanius tigrinus, Yellowrumped Flycatcher Ficedula zanthopygia and Mugimaki Flycatcher F. mugimaki. These species are easily identified and, though not scarce, are sufficiently noteworthy to be recorded by amateur observers, yielding a reasonably large dataset. |
||||||||||||||||||||||||||||||||||||||
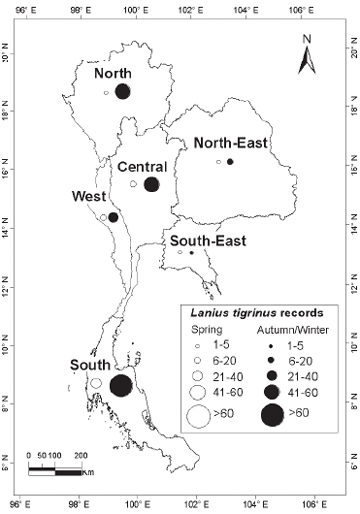
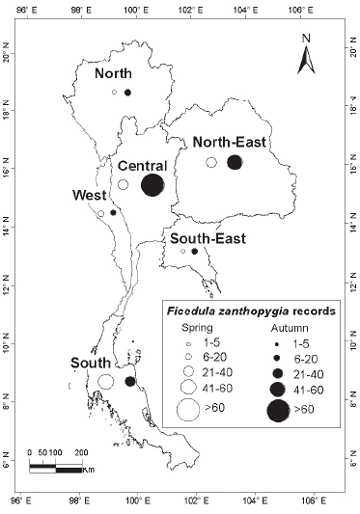
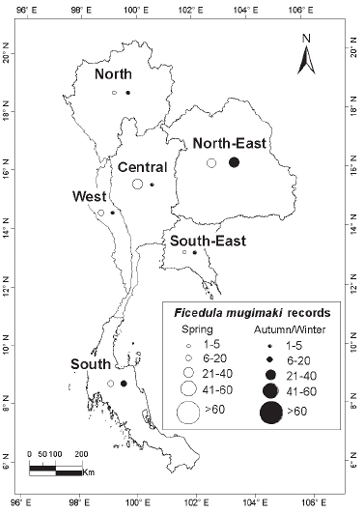
| METHODS The principal sources of data were records submitted by local or visiting birdwatchers from 1980 onwards; my own sight records and mist-net captures; published records or major compilations where dates and localities were given (e.g. Robinson & Kloss 1921–1924, Riley 1938, Deignan 1945, Meyer de Schauensee 1946); the small number of specimens held in the Centre for Thai National Reference Collections, Environment and Resources Department, Thailand Institute of Scientific and Technological Research, Bangkok, and in the Boonsong Lekagul collection stored in the National Science Museum, Bangkok. Although specimens and ringing records (mainly from Laem Phak Bia, a coastal site in the Gulf of Thailand: Round & Kongtong 2009) distinguished among sex and age classes of the three taxa, these contributed only a small proportion of records, most of the rest being sightrecords. For the two flycatchers, sight-records distinguished only between birds in definitive adult male body plumage (with black upperparts) and ‘others’ (brown-plumaged, female/immature individuals). While male and female Tiger Shrikes in spring differ in plumage (females with much reduced black on forehead: Wells 2007), few field observers distinguished between the sexes. Additionally, Tiger Shrike is unusual in that adults have two complete moults per year (Prys-Jones 1991). Adults undergo a post-nuptial moult on the breeding grounds into a brown, juvenile-like plumage, so that many autumn adults are difficult to separate reliably from juveniles. Accordingly, in this analysis no distinction was made between sexes or age-classes in Tiger Shrike, while the two flycatchers’ age and sex classes were given as definitive males and ‘others’. Records for each species were grouped by ten-day period throughout the year in order to standardise the nationwide pattern of occurrence. Maps were used to illustrate the geographical scatter of records, based on the six regions first proposed by Kloss (1915) and subsequently depicted in King et al. (1975): North (alternative name North-West), North-East, South-East, West (South-West), Central and South (Peninsula). |
||||||||||||||||||||||||||||||||||||||
| RESULTS Over 720 records for the three species were subjected to analysis. Although these covered a span of years from 1896 to the present, more than 90% of the records for each of the three species post-dated 1980 (Fig. 1). Although initially it was assumed that the overwhelming majority of records would be from the Central Region, which includes the city of Bangkok, where most observers are concentrated, sightings from the Central Region only contributed 39.7% of records of the three species combined. A major surprise was the paucity of records from the Northern Region (only 4.4% of records of all three species), even though this includes Chiang Mai, Thailand’s second largest city, which supports many resident birdwatchers. The North-East, the largest region in terms of land area, was expectedly little-watched, with the exception of Khao Yai National Park (at the extreme south-west of the region, and the major source of records). |
||||||||||||||||||||||||||||||||||||||
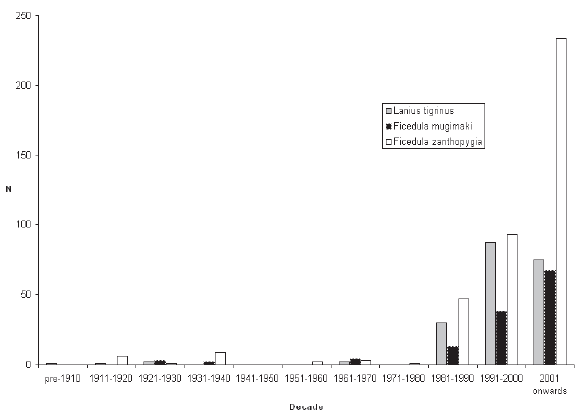 Figure 1. Records of three study species according to decade. |
||||||||||||||||||||||||||||||||||||||
| Tiger
Shrike A total of 202 records involving 324 individual birds was compiled (Fig. 2), of which 259 were in autumn and 56 in spring. Only nine individuals (2.8%) were from the midwinter period (November to mid-March), indicating that this species was almost exclusively a spring and autumn passage migrant. The earliest autumn record was 11 August and the latest 18 October, though almost all (98.8%) occurred before 10 October (median date 13 September: Table 1). Although the peak autumn passage period overall was the second week of September, disproportionately more of those after the first ten days of September were from the South, reflecting a geographical shift in the population. The peak period of occurrence around Bangkok and elsewhere in continental Thailand appeared to be during the last week of August and the first week of September. |
||||||||||||||||||||||||||||||||||||||
Table
1. Occurrence of Tiger Shrike Lanius tigrinus,
Yellow-rumped Flycatcher Ficedula zanthopygia and Mugimaki
Flycatcher F.mugimaki on
|
||||||||||||||||||||||||||||||||||||||
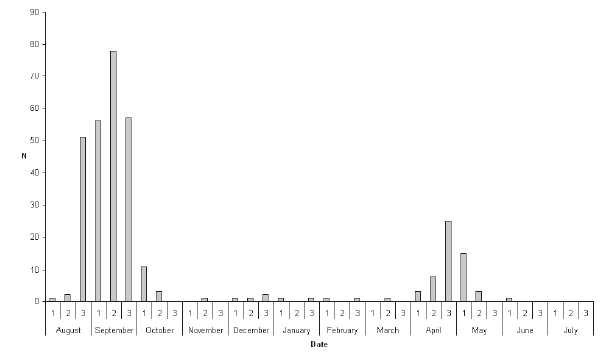
Figure 2. Seasonal distribution of records of Tiger Shrike Lanius tigrinus by ten-day period. |
||||||||||||||||||||||||||||||||||||||
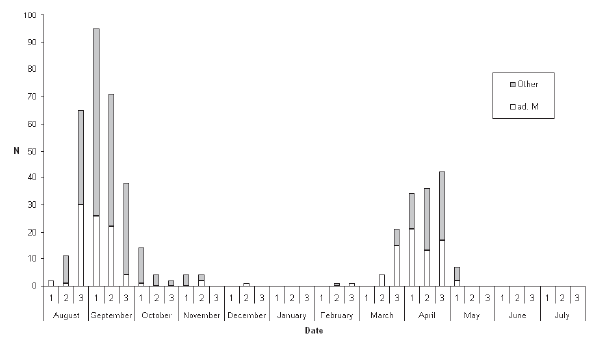 Figure 3. Seasonal distribution of records of Yellow-rumped Flycatcher Ficedula zanthopygia by ten-day period. |
||||||||||||||||||||||||||||||||||||||
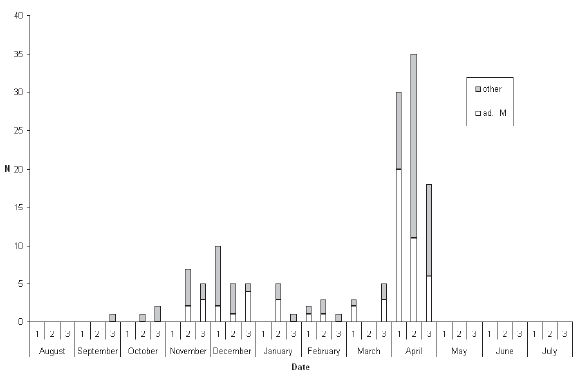 Figure 4. Seasonal distribution of records of Mugimaki Flycatcher Ficedula mugimaki by ten-day period. |
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
| The
earliest apparent spring passage record was 12 March, although since
no others were recorded until 6 April there is a possibility that
this was an aberrant wintering bird. The latest date was 6 June, although
the peak spring passage was in the last ten days of April and the
median date was 30 April (Table 1). For both passage seasons combined proportionately more records were obtained from the South (46.3%) relative to the Central Region (27.9%) than in the other two species examined (Fig. 5). Additionally, five of the nine midwinter records of Tiger Shrikes were from the South, suggesting the possibility that a small population may genuinely overwinter in the provinces adjacent to the Malaysian border. Yellow-rumped Flycatcher The dataset comprised 395 records of 457 individuals. This species was similarly almost exclusively a spring and autumn passage migrant, with only seven individuals (1.5%) recorded during the midwinter period, from mid- November to mid-March. More than twice as many individuals were recorded in autumn (306) than in spring (144). Autumn passage was recorded from 1 August onwards to early November, although 71% of all autumn records were in the last ten days of August and the first twenty days of September, and only 20 individuals (6.5% of autumn records) were obtained after the beginning of October (Fig. 3). Spring passage was recorded from 19 March to 9 May (Table 1), with over three-quarters of those sighted during March being male (Fig. 3). Most observers did not distinguish between brown-primaried first-year males and black-primaried adult males. The median date for spring males (10 April) was one week earlier than that for females (17 April; Table 1). Adult males also constituted a significantly higher proportion of earlier-arriving birds in autumn: 42.3% of August birds were adult males, compared with only 25.5% of September birds (?2 = 7.44; p < 0.01; Fig. 3). The median date for adult males in autumn was 4 September compared with 10 September for other age/sex-classes combined (Table 1). Four of seven midwinter birds were black-bodied males. The winter sample is too small to draw any firm conclusions and does not necessarily imply that proportionately more adult males overwinter, since some could have been first-winters that had completed their body moult. The proportion of black body-plumaged males in spring was exactly 50%, all first-year males having by then acquired black upperparts. Fifty percent of records of Yellow-rumped Flycatchers were from the Central Region compared with only 21.2% from the South (Fig. 6). Records from the South did not figure disproportionately among the few Yellow-rumped Flycatchers recorded in midwinter, so there was no evidence of a significant wintering population anywhere, including in the extreme southernmost provinces, even though Yellow-rumped Flycatcher winters in Malaysia (Wells 2007). |
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
| DISCUSSION All three species considered have breeding ranges in the Eastern Palaearctic, with the smallest range being found in Tiger Shrike, breeding mainly in Ussuriland, the Korean Peninsula, Japan and north-east China (Brazil 2009). Two of the three breed in smaller woodlots and parks (including in urban areas: J. W. Duckworth in litt.) in addition to forest. The third, Mugimaki Flycatcher, is the most restricted to forest on the breeding grounds (Brazil 2009) and also tends to be more restricted to taller woodland on passage in South-East Asia than the other two. Although both Tiger Shrike and Yellow-rumped Flycatcher winter commonly in Malaysia (Wells 2007), neither does so regularly or widely in peninsular Thailand (apart from a few Tiger Shrikes that possibly winter in the extreme southern provinces, immediately adjacent to the Malaysian border). Mugimaki Flycatcher is a winterer and passage migrant in both seasons in Malaysia but commonest above the montane ecotone (Wells 2007). In Laos this species is also mainly associated with hill-slope and montane habitats, both on passage and in winter (Duckworth et al. 1998, J. W. Duckworth in litt.). It is highly likely, therefore, that many more might be recorded during winter if the little-covered southern Thai mountains were more accessible. As passage migrants, Tiger Shrike and Yellow-rumped Flycatcher showed overall many more records in autumn than in spring, while the reverse was true in Mugimaki Flycatcher. The relative scarcity of both Tiger Shrike and Yellow-rumped Flycatcher in spring is unlikely to be an artifact of coverage as, in general, more birdwatchers and bird photographers are searching for birds at that time than in autumn. Some other migrant shrikes (e.g. the Western Palaearctic-breeding, African-wintering Red-backed Shrike L. collurio and Lesser Grey Shrike L. minor) are noted ‘loop migrants’, and take a more easterly route on northward (spring) migration than on southward (autumn) migration (Moreau 1961, 1972). There is no obvious reason, however, why there should be any parallel among the Eastern Palaearctic–South-East Asian migrant species. Additionally, Tiger Shrike is very rare in Hong Kong, with only nine records, all of which were in autumn (Carey et al. 2001), so it is unlikely that spring migrants take a more easterly northwards route. Nor are there many records to the west (see below). Because Tiger Shrike is a relatively late spring migrant, with northwards passage extending well into May (latest Thai-Malay Peninsula date 17 May: Wells 2007) some may be overlooked. The most likely explanation, however, is that most northbound spring migrants, having fattened in Malaysian or Indonesian wintering areas, overfly Thailand (and southern China). This interpretation is corroborated by the scarcity of records from Laos in spring, even though coverage by visiting birdwatchers in Laos probably reaches its annual peak at that time (J. W. Duckworth in litt.). The depiction of differing geographical distributions among the three species is still tentative as even now some regions (especially the South-East) are much less-visited than others, and would surely contribute many more records if better covered. In the case of Yellow-rumped Flycatcher the disparity between the 50% of records from the Central Plains region compared with only 21.2% from the South may to some extent be due to a proportionate disparity of (especially autumn) coverage between the two regions. However, Wells (2007) suggested that the species was less common in the Thai peninsular provinces in autumn than in spring, and this is borne out by the present analysis. The apparent scarcity of Yellow-rumped Flycatcher records from the North (which contributed just 2.8% of records for both seasons combined) might be partly due to inadequate coverage of favoured lowland garden or parkland habitats in that (mainly hilly or mountainous) region. But such a pattern might also occur if Yellow-rumped Flycatcher had a slightly more north-east/southwest migratory orientation that caused it largely to miss the North on both migrations. North-East Thailand contributed 15.3% of spring records, while Yellow-rumped Flycatcher is also regular in Laos in spring (J. W. Duckworth in litt.). This would tend to rule out spring overflight as a cause for scarcity in the North. In Tiger Shrike by comparison, overflight is a more likely explanation as there were equivalently few spring records from both the North and North-East. The Yellow-rumped Flycatcher in Hong Kong has a pattern of occurrence similar to that of Tiger Shrike. It is very scarce in spring (only one record: Carey et al. 2001), suggesting that it either overflies southern China or follows a route that bypasses the coast. Although more frequent in autumn, it is still much less common there than in Thailand. The peak passage in Hong Kong, in mid- September, more or less corresponds with that 8º latitude further south, in Thailand’s Central Region. In autumn, Mugimaki Flycatcher was a much later migrant, with passage peaking over one month later than the other two. This made it difficult to distinguish between migrants and winterers. The late timing of passage in this species is also corroborated from night-time interceptions in Malaysia (Wells 2007). The predominance of spring passage records almost certainly accurately reflects a genuine greater abundance then, especially since the chances of encountering any individual passage migrant should be lower in spring than in autumn. Not only is the total population of any given Palaearctic migrant in spring smaller than that in autumn, owing to winter mortality, but spring migrants in general may also make shorter migration stop-overs, owing to the imperative to return to the breeding grounds to take up territory. In Hong Kong, where the Mugimaki Flycatcher is similarly a scarce winter visitor and passage migrant, it is also recorded more commonly in spring than in autumn (Carey et al. 2001). In general, the broad correspondence between Thailand and Hong Kong in records of the three species gives no reason to speculate that autumn and spring migratory routes differ in major respects for any of them, other than in possible spring overflight of northern South- East Asia and southern China. Nor does any species pass much to the west in either season. Yellow-rumped Flycatcher is a vagrant to peninsular India, while neither Tiger Shrike nor Mugimaki Flycatcher have been recorded there (Rasmussen & Anderton 2005). In Burma, Yellowrumped Flycatcher and Tiger Shrike are known from the extreme east of the country and Tenasserim only, while there are no records of Mugimaki Flycatcher (Robson 2008). The Mugimaki Flycatcher showed a generally more eastern distribution in Thailand than either Tiger Shrike or Yellow-rumped Flycatcher. Well-watched forest sites in Western Thailand, such as Kaeng Krachan National Park, which covers a similar altitudinal range to Khao Yai, have yielded many fewer records than the latter site, while there were only two records from the very heavily watched mountains in Chiang Mai Province, in the western part of the North. Definitive male Mugimaki Flycatchers accounted for 42% of spring records, proportionately fewer than in Yellow-rumped Flycatcher. Although this might be expected, since male Mugimaki Flycatchers may not attain black upperparts until their second year, the difference in the proportions of recognisable males was not statistically significant. Although the numbers of all three migrant species recorded increased markedly after 1980, owing to greatly increased coverage by birdwatchers and to better collation of records, the numbers of Tiger Shrikes recorded did not increase in proportion to those of Yellow-rumped Flycatchers, and indeed there were fewer Tiger Shrike records post-2000 than in the preceding decade (Fig. 1). Given the fact that the peninsula contributed most records of Tiger Shrike overall, one possible reason for this disparity might be that coverage increased more in the Central Region post-2000 than it did in the South (where either coverage, or record submission, may have actually declined). Another possibility is that the decline in records reflects an ongoing global decline in numbers of Tiger Shrike that was reported by BirdLife International (2009). This analysis shows how records collected mainly by amateur birdwatchers may make a contribution to the scientific record in Thailand and South-East Asia as they have long done in Europe and North America. Although the present paper has drawn on records spanning a roughly 110-year period, over 80% of the records of each species post-dated 1990 and therefore provided a reasonable ‘snapshot’ of present patterns. Studies on the phenology of breeding and migration have assumed added urgency owing to the weight of evidence that climate change is having major impacts on birds and biological systems (Crick & Sparks 1999, Butler 2003, Coppack & Both 2003, Sanderson et al. 2006). Migrant birds may be at elevated risk from climate change because their annual cycles are adapted to cope with the vicissitudes of climate in widely separated wintering and breeding areas, which may differ in the extent to which they are affected by climate change. Additionally, clearance of forest for agriculture has impacted the status of both resident and migrant birds, removing large swathes of habitat for inhabitants of forest, while simultaneously opening up huge areas for those that favour open country. Wells (2007) has documented a gradual southwards expansion of the winter range of migratory Black Drongos Dicrurus macrocercus, an open-country winterer, in Malaysia, and range shifts among a suite of other migrant species might be expected. Migrant birds additionally face a number of other threats including direct persecution, and indirect disturbance. Mortality during nocturnal migratory flights due to collisions and disorientation caused by communications towers, other man-made structures, and gas flares from offshore oil-fields has also been demonstrated and could cause population declines (Lid 1977, Morris et al. 2003). Improved understanding of migrant bird ecology, and a capacity to monitor changes in the numbers and distribution of migrant birds, are therefore of great importance. Although this paper establishes a rough baseline on seasonality and distribution for the three species considered, more intensive and systematic monitoring, from a greater and more even spread of sites, for a greater range of species, and comparing seasonal patterns decade by decade, or by five-year period, into the future would refine and improve understanding. This could be done through better collation of records from local birdwatching groups (several of which already exist for the Thai regions), university bird clubs, and possibly in future even formally established bird observatories, including those in neighbouring countries. Round & Kongtong (2009) recommended the establishment of a bird observatory at one coastal site, the Laem Phak Bia Environmental Research and Development Project, Phetchaburi, where ringing has already been implemented for a decade, and which contributed a number of records to the present analysis. |
||||||||||||||||||||||||||||||||||||||
| ACKNOWLEDGEMENTS I am grateful to the many birdwatchers and bird photographers who contributed their sightings to this analysis. I should also like to thank Des Allen, Robert DeCandido, Will Duckworth, David Pearson, Andrew Pierce, Stephen Rumsey and David Wells for their comments on drafts of this paper. I am grateful to the staff and director of the Laem Phak Bia Environmental Research and Development Project for permission to ring birds there. I thank the directors of the Thailand Institute of Scientific and Technological Research and the National Science Museum, Thailand, for permitting access to specimens. I also thank Niti Sukumal and Supatcharee Tanasarnpaiboon for kindly preparing the distribution maps. My work is supported by The Wetland Trust (UK). |
||||||||||||||||||||||||||||||||||||||
| REFERENCES BirdLife International (2009) Species factsheet: Lanius tigrinus. Downloaded from http://www.birdlife.org on 21/2/2010. Brazil, M. (2009) Birds of East Asia. London: Christopher Helm. Butler, C. J. (2003) The disproportionate effect of global warming on the arrival dates of short-distance migratory birds in North America. Ibis 145: 484–495. Carey, G. J., Chalmers, M. L., Diskin, D. A., Kennerley, P. R., Leader, P. J., Leven, M. R., Lewthwaite, R. W., Melville, D. S., Turnbull, M. & Young, L. (2001) The avifauna of Hong Kong. Hong Kong: Hong Kong Birdwatching Society. Chalmers, M. L. (1986) Annotated checklist of the birds of Hong Kong. Fourth edition. Hong Kong: Hong Kong Bird Watching Society. Coppack, T., & Both, C. (2003) Predicting life-cycle adaptation of migratory birds to global climate change. Ardea 90: 369–378. Crick, H. P. & Sparks, T. H. (1999) Climate change related to egglaying trends. Nature 399: 423–424. Deignan, H. G. (1945) The birds of northern Thailand. US Natn. Mus. Bull. 186. Duckworth, J. W., Tizard, R. J., Timmins, R. J., Thewlis, R. M., Robichaud, W. B. & Evans, T. D. (1998) Bird records from Laos, October 1994–August 1995. Forktail 13: 33–68. King, B., Dickinson, E. C. & Woodcock, M. W. (1975) Field guide to the birds of South-East Asia. London: Collins. Kloss, C. B. (1915) Zoo-geographical divisions for Siam. J. Nat. Hist. Soc. Siam 1: 250–251. Lekagul, B. & Round, P. D. (1991) A guide to the birds of Thailand. Bangkok: Saha Karn Bhaet. Lid, G. (1977) Fugler brennes ihjel av gassflaminer i Nordsjøen. Fauna 30: 185–190. Meyer de Schauensee, R. (1946) On Siamese birds. Proc. Acad. Nat. Sci. Philadelphia 93: 1–82. Moreau, R. E. (1961) Problems of Mediterranean-Saharan migration. Ibis 103a: 373–427, 580–623. Moreau, R. E. (1972) The Palaearctic-African bird migration systems. London: Academic Press. Morris, S. R., Clark, A. R., Bhatti, L. H. & Glasgow, J. L. (2003) Television tower mortality of migrant birds in western New York and Youngstown, Ohio. Northeastern Naturalist 10: 73–76. Prys-Jones, R. P. (1991) The occurrence of biannual primary moult in passerines. Bull. Brit. Orn. Club 111: 150–152. Rasmussen, P. C. & Anderton, J. C. (2005) Birds of South Asia: the Ripley guide. Washington, D.C.: Smithsonian Institution. Riley, J. H. (1938) Birds from Siam and the Malay Peninsula in the United States National Museum collected by Drs. Hugh M. Smith and William L. Abbott. US Natn. Mus. Bull. 172. Robinson, H. C. & Kloss, C. B. (1921) The birds of south-west and peninsular Siam. J. Nat. Hist. Soc. Siam 5: 1–397. Robson, C. (2008) A field guide to the birds of South-East Asia. London: New Holland. Round, P. D. & Kongtong, W. (2009) Birds of Laem Phak Bia. Bangkok: The Chaipattana Foundation. Sanderson, F. J., Donald, P. F., Pain, D. J., Burfield, I. J. &. van Bommel, F. P. J. (2006) Long-term population declines in Afro- Palaearctic migrant birds. Biol. Conserv. 131: 93–105. Sharrock, J. T. R. (1974) Scarce migrant birds in Britain and Ireland. Berkhamsted: T. & A. D. Poyser. Wells, D. R. (2007) The birds of the Thai-Malay Peninsula, 2. Passerines. London: Christopher Helm. |
||||||||||||||||||||||||||||||||||||||
| Kindly
submitted by: Philip D. Round, Assistant Professor and Regional Representative, The Wetland Trust, Department of Biology, Faculty of Science, Mahidol University, Rama 6 Road, Bangkok 10400, Thailand. Email: frpdr@mahidol.ac.th |
||||||||||||||||||||||||||||||||||||||
Tweet

|
||||||||||||||||||||||||||||||||||||||

A Guide to Birdwatching in Thailand. Copyright © 2004-2017 thaibirding.com. All rights reserved.